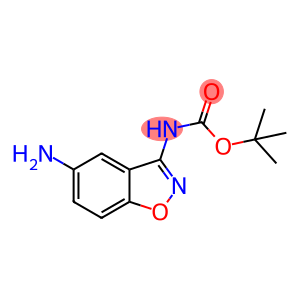
Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI)
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate
CAS: 380629-73-2
Molecular Formula: C12H15N3O3
Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI) - Names and Identifiers
| Name | tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate |
| Synonyms | LogP tert-butyl 5-amino-1,2-benzisoxazol-3-ylcarbamate tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate tert-Butyl (5-aMinobenzo[d]isoxazol-3-yl)carbaMate (5-AMinobenzoisoxazol-3-yl)carbaMic acid tert-butyl ester (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER CarbaMic acid, (5-aMino-1,2-benzisoxazol-3-yl)-, 1,1-diMethylethyl ester Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI) |
| CAS | 380629-73-2 |
| InChI | InChI=1/C12H15N3O3/c1-12(2,3)17-11(16)14-10-8-6-7(13)4-5-9(8)18-15-10/h4-6H,13H2,1-3H3,(H,14,15,16) |
Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI) - Physico-chemical Properties
| Molecular Formula | C12H15N3O3 |
| Molar Mass | 249.27 |
| Density | 1.306±0.06 g/cm3(Predicted) |
| Boling Point | 384.7±22.0 °C(Predicted) |
| Flash Point | 186.4°C |
| Vapor Presure | 4.02E-06mmHg at 25°C |
| pKa | 11.73±0.43(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.638 |
Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI) - Introduction
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate (also known as tert-Butyl -1,2-benzoxazol-3-yl)carbamate) is an organic compound. The following is a detailed description of its nature, use, formulation and safety information:
Nature:
-Appearance: White solid
-Molecular formula: C12H16N2O3
-Molecular weight: 236.27g/mol
-Melting point: 117-119°C
-Boiling point: 460.1°C
-Solubility: Soluble in a variety of organic solvents, such as ethanol and dimethylformamide (DMF)
-Stability: The compound is relatively stable under normal storage conditions.
Use:
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate is often used as an intermediate in organic synthesis. It can be used in the synthesis of other organic compounds, such as pharmaceuticals and dyes. It can also be used as a bactericide, desulfurizer and antioxidant.
Preparation Method:
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate can be synthesized by the following steps:
1. Synthesis of 5-chloro-1, 2-benzoxazole:
-Take 1,2-benzo (D) isoxazole and thionyl chloride to react to obtain 5-chloro-1, 2-benzoxazole.
2. Synthesis of 5-aminobenzo [D] isoxazole:
-5-chloro-1, 2-benzoxazole is reacted with ammonia water under alkaline conditions to obtain 5-aminobenzo [D] isoxazole.
3. The reaction produces the final product:
-React 5-aminobenzo [D] isoxazole with tert-butyryl chloride, and then with tert-butanol to form tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate.
Safety Information:
When manipulating and using tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate, general laboratory safety practices should be followed. It can be irritating and toxic, so wear appropriate personal protective equipment such as gloves and goggles. Care should be taken to avoid contact with skin, eyes and inhalation of gases or dust. If accidental contact occurs, immediately flush the affected area with water and seek medical help.
Nature:
-Appearance: White solid
-Molecular formula: C12H16N2O3
-Molecular weight: 236.27g/mol
-Melting point: 117-119°C
-Boiling point: 460.1°C
-Solubility: Soluble in a variety of organic solvents, such as ethanol and dimethylformamide (DMF)
-Stability: The compound is relatively stable under normal storage conditions.
Use:
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate is often used as an intermediate in organic synthesis. It can be used in the synthesis of other organic compounds, such as pharmaceuticals and dyes. It can also be used as a bactericide, desulfurizer and antioxidant.
Preparation Method:
tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate can be synthesized by the following steps:
1. Synthesis of 5-chloro-1, 2-benzoxazole:
-Take 1,2-benzo (D) isoxazole and thionyl chloride to react to obtain 5-chloro-1, 2-benzoxazole.
2. Synthesis of 5-aminobenzo [D] isoxazole:
-5-chloro-1, 2-benzoxazole is reacted with ammonia water under alkaline conditions to obtain 5-aminobenzo [D] isoxazole.
3. The reaction produces the final product:
-React 5-aminobenzo [D] isoxazole with tert-butyryl chloride, and then with tert-butanol to form tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate.
Safety Information:
When manipulating and using tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate, general laboratory safety practices should be followed. It can be irritating and toxic, so wear appropriate personal protective equipment such as gloves and goggles. Care should be taken to avoid contact with skin, eyes and inhalation of gases or dust. If accidental contact occurs, immediately flush the affected area with water and seek medical help.
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Visit Supplier Webpage Request for quotationCAS: 380629-73-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Request for quotation
CAS: 380629-73-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 380629-73-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate Request for quotation
CAS: 380629-73-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 380629-73-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Visit Supplier Webpage Request for quotation
CAS: 380629-73-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 380629-73-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Visit Supplier Webpage Request for quotationCAS: 380629-73-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Request for quotation
CAS: 380629-73-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 380629-73-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: tert-Butyl (5-amino-1,2-benzoxazol-3-yl)carbamate Request for quotation
CAS: 380629-73-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 380629-73-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: (5-AMINOBENZO[D]ISOXAZOL-3-YL)CARBAMIC ACID TERT-BUTYL ESTER Visit Supplier Webpage Request for quotation
CAS: 380629-73-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 380629-73-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Carbamic acid, (5-amino-1,2-benzisoxazol-3-yl)-, 1,1-dimethylethyl ester (9CI)
4H-1-Benzopyran-4-one, 2,3-dihydro-5-hydroxy-3-(4-hydroxy-2-methoxyphenyl)-7-methoxy-
4H-1-Benzopyran-4-one, 2,3-dihydro-5-hydroxy-3-(4-hydroxy-2-methoxyphenyl)-7-methoxy-



